Type 2 diabetes is diagnosed based on blood values – but the actual changes take place in many organs, including the tissue of the pancreas, where insulin is produced. However, these changes are not visible to the human eye under a microscope. A research team from the German Diabetes Center (DDZ) in Düsseldorf, the Paul Langerhans Institute of Helmholtz Munich at the Carl Gustav Carus University Hospital of the Technical University of Dresden (PLID), and the Institute for Diabetes Research and Metabolic Diseases at Tübingen University Hospital – three partner institutes of the German Center for Diabetes Research (DZD) – has now shown for the first time that Explainable artificial intelligence can now distinguish which tissue comes from people with type 2 diabetes and which structures play a role in this process. This is based on a cross-location data set from Dresden and Tübingen. Researchers from the German Cancer Research Center (DKFZ) and Helmholtz Imaging were also involved.
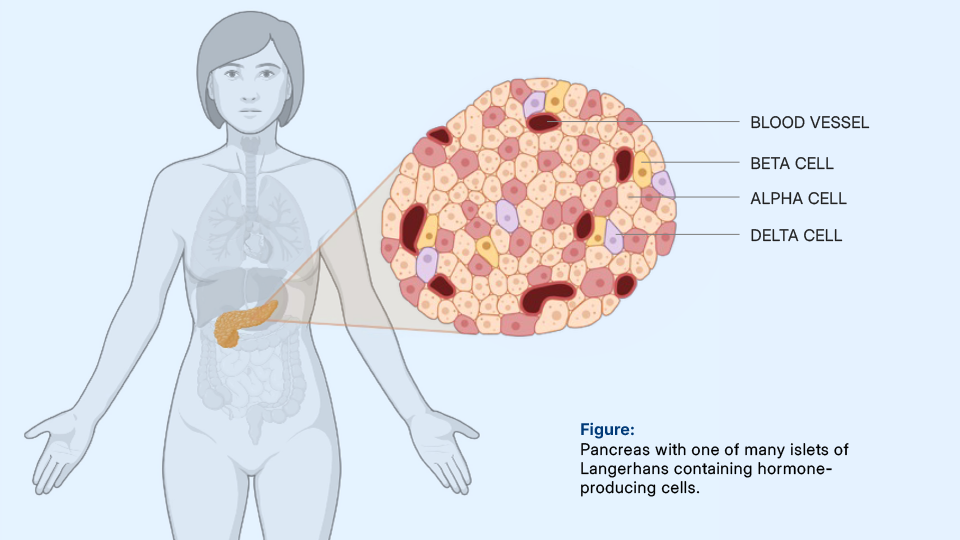
Type 2 diabetes is one of the most common chronic diseases worldwide. It is usually diagnosed on the basis of elevated blood glucose levels. However, these levels primarily reflect the consequences of the disease rather than its structural causes. The key changes take place in the pancreas. In the so-called islets of Langerhans in the pancreas, specialized cell clusters produce hormones that regulate blood glucose. Until now, research has focused primarily on beta cells, whose loss of function is considered a decisive step in the development of type 2 diabetes.
However, alpha cells, which produce the blood glucose-raising hormone glucagon, and delta cells, which slow down the release of insulin and glucagon, are also part of this finely tuned system. In addition, there are blood vessels, connective tissue, fat cells, and nerve fibers, which together influence the function of the islets. Whether and how this complex interaction changes structurally in type 2 diabetes is difficult to assess at this stage. Under the microscope, the differences within the islets of Langerhans are often barely discernible, partly because cell composition and arrangement vary greatly from person to person.
Explainable AI reveals hidden changes in type 2 diabetes
This is where the current study by researchers from the German Diabetes Center (DDZ), the Paul Langerhans Institute at Dresden University Hospital, the Institute for Diabetes Research and Metabolic Diseases at Tübingen University Hospital, and the German Cancer Research Center comes in. “With 100 samples from Dresden and Tübingen, we have built up one of the most comprehensive data sets to date from pancreatic tissue sections,” explains Dr. Felicia Gerst, a scientist at the Institute for Diabetes Research and Metabolic Diseases at the University Hospital of Tübingen.
The samples come from people who have undergone pancreatic surgery for various medical reasons. Machine learning was used to train a model that can distinguish between large, information-rich tissue sections from people with and without type 2 diabetes with a high degree of accuracy and predict diabetes status. Crucially, the system works in an explainable way. It marks the areas of the image that influence the decision so that they can then be biologically evaluated and statistically tested.
Changes are not limited to individual cell types
The analyses showed that it is not only beta cells that change in type 2 diabetes. Alpha and delta cells also occur in different positions and distributions. In addition, the islets of Langerhans are smaller overall. Another important finding concerns fat cells in the pancreas. In people with type 2 diabetes, fat cells are more frequently found in the immediate vicinity of the islets of Langerhans. “This proximity could influence the function of the hormone-producing islets of Langerhans,” explains Professor Robert Wagner, head of the Clinical Study Center working group at the German Diabetes Center (DDZ). “The findings provide us with concrete starting points for further research. Because if we understand the structural mechanisms of the disease and know which processes drive it, we can develop new therapies in a more targeted manner,” adds Wagner, who initiated the project.
“The research project shows that we need to look not only at individual cells, but at the entire microscopic network of the pancreas in order to better understand diabetes,” emphasizes Michele Solimena, Professor of Molecular Diabetology at Dresden University Hospital and spokesperson for the Paul Langerhans Institute Dresden. In their publication, the researchers provide the complete code for image preprocessing, model training, and explanatory analyses. In doing so, they create transparency and enable other working groups to further develop the method and apply it to other questions.
Original publication:
Titel: Explainable AI-based analysis of human pancreas sections identifies traits of type 2 diabetes
Journal: Nature Communications
Autoren: Klein, L., Ziegler, S., Gerst, F., Morgenroth, Y., et al.
Doi: https://doi.org/10.1038/s41467-026-69295-2